Information
Journal Policies
C-DNA Content and Chromatin Distribution in Diploid and Large Bovine Sperm Nuclei
Maria R. Ferrari1 , Liliana O. Gonzalez1 , Susana M. Giuliano1 , Susana H. Campi1 , Maria L. Fischman1, Humberto O. Cisale1*
Copyright :© 2018 Authors. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
The aims of the present work where: i) to determine and compare the DNA content and parameters of chromatin distribution in normal, large and diploid bovine sperm nuclei, ii) to evaluate the frequency of these three classes of nuclei, iii) and to analyze the usefulness of the Feulgen reaction to recognize normal, large and diploid sperm nuclei in bovine ejaculates. Cytophotometric determinations showed that the C-DNA values of normal and large nuclei were not significantly different but differed statistically (P< 0.05) from those of diploid nuclei. Cytophotometric parameters related to chromatin distribution showed statistical differences between normal, large and diploid nuclei (P< 0.05). The Area and Volume of large sperm nuclei were greater than those of normal nuclei, whereas the Mean and Maximal Absorbance were smaller, suggesting incomplete and/or defective chromatin condensation in large nuclei. Diploid nuclei showed an anisotropic DNA increase with a privileged direction of growth in the perpendicular plane of the principal area of the sperm nucleus. The Feulgen reaction stained the three classes of nuclei with different intensity, allowing us to recognize them clearly using light microscopy. The frequencies of large and diploid bovine sperm nuclei were 0.27% and 0.08% respectively. The morphological description of diploid nuclei differs between authors. We propose that these differences could be the consequence of failures in different pre-meiotic or meiotic stages.
Bovine, C-DNA, Diploid Sperm Nuclei, Feulgen Reaction, Large Sperm Nuclei, Sperm Chromatin.,Urology
1. Introduction
Diploid and large sperm nuclei have been described in different mammalian species as abnormalities that can be related to fertility problems (Gledhill, 1965; Egozcue et al., 2000; Levek-Motola et al., 2005; Spirito et al., 2011). Morphological descriptions and interpretations of their origin differ between different authors (Ferrari et al., 1998; Celik-Ozenci et al., 2004; Vieytes et al., 2008; Revay et al., 2009; Spirito et al., 2011). Although the frequency of diploid spermatozoa is usually low in semen of fertile individuals, these spermatozoa can contribute to the production of triploid embryos and are therefore one of the causes of spontaneous abortion (Egozcue et al., 2002; Codina-Pascual et al., 2006; Rosenbusch, 2008). Diploid spermatozoa are the result of various abnormalities that may occur before meiosis or during the first and/or the second meiotic division (Egozcue et al., 2000; Codina-Pascual et al., 2006).
Chemical and architectural modifications of chromatin (DNA–protein complex) may lead to a disturbed condensation during spermatogenesis, which could be the origin of large nuclei. These nuclei are usually wider and longer than normal and are associated with low fertility or even total infertility (Barth & Oko, 1989; Ferrari et al., 1998; Revay et al., 2009; Chemes & Rawe, 2012; Guthauser et al., 2016).
Different methods have been used to evaluate the ploidy level of sperm nuclei: biochemical methods (Pogany et al., 1981), microspectro photometric measures of Feulgen-stained nuclei (Gledhill, 1965; Vieytes et al., 2008; Spirito et al., 2011). FISH using specific probes to recognize chromosomes (Celik-Ozenci et al.,2004) and flow cytometry (Levek-Motola et al., 2005).
The size and shape of sperm heads and nuclei are important parameters for semen evaluation and when they differ from the normal pattern for the species, the fertility of the ejaculates can be reduced and abortions may occur (Chemes & Rawe, 2003; Enciso et al., 2011). Therefore, knowledge of morphometric and morphological parameters of diploid and large spermatozoa become important to evaluate the ejaculate quality and implement assisted reproduction techniques.
The aims of the present work were: i) to determine the DNA content and parameters of chromatin distribution in bovine sperm nuclei to compare normal, large and diploid nuclei; ii) to evaluate the frequency of these three classes of nuclei in bovine ejaculates; iii) and to discuss the usefulness of the Feulgen reaction, a DNA-specific and stoichiometric stain to recognize normal, diploid and large sperm nuclei in bovine ejaculates.
2. Materials And Methods
We analyzed 35 doses of frozen semen straws from 35 bulls. The fertility potential was estimated into three fertility categories according to the conception rates (CR) (high fertility: CR higher than 50%; medium fertility: CR between 40% and 50% and low fertility: CR lower than 40%). The bulls were treated in accordance with established ethical guidelines.
The Feulgen reaction was performed according to the technique already described for bovine spermatozoa (Vieytes et al., 2008). This reaction is specific and stoichiometric with the DNA.
Semen smears were stained with the Feulgen reaction and analyzed under light microscopy (x1000). All observations were made on the flattening plane by the same trained operator. Normal, diploid and large nuclei were classified taking into account the staining intensity and their size and shape. The morphology of normal and large nuclei was determined according to the classification scheme of Barth & Oko (1989) and the diploid nuclei according to the descriptions of different authors (Gledhill, 1965; Ferrari et al., 1998; Vieytes et al., 2008).
Feulgen densitometry relies on the premise that the amount of stain bound is directly proportional to the amount of DNA present and the amount of stain is determined based on the amount of light it absorbs. According to Hardie et al., (2002), the Lambert’s and Beer’s law holds for measurements of stained nuclear DNA. Measurements were made in semen of five fertile males at a wavelength of 560 nm using a Cytoscan Zeiss Universal microscope (UMSP 30) and X2000. For each nucleus, Area, Maximal absorbance (MaxAbs), Mean absorbance (MeAbs) and Mean absorbance per area were determined. The latter corresponded to the DNA content expressed in arbitrary units. The C-value (expressed in picograms) was calculated using erythrocyte DNA from Gallus domesticus as standard (C-value = 1.25 pg) (Gregory, 201(8). The Volume (expressed in arbitrary units) was inferred from the product between Area and MeAbs. All measurements were made by the same operator.
SURFER Version Sep. 12 2012 Golden Software, Inc. Point was used to construct 2D surface maps.
The percentage of coincidence was calculated using the following expression: (Number of cases where optical analysis and cytophotometric classification coincided / total number of sperm nuclei analyzed) x 100.
The normality of the cytophotometric data was tested with the Shapiro-Wilk Test. Differences between the three classes of sperm nuclei (normal, diploid and large) were evaluated using the Kruskal Wallis Test. The statistical analyses were performed using the Infostat software, Facultad de Ciencias Agrarias, Universidad Nacional de Córdoba, Córdoba, Argentina (Di Rienzo et al., 2015).
3. results
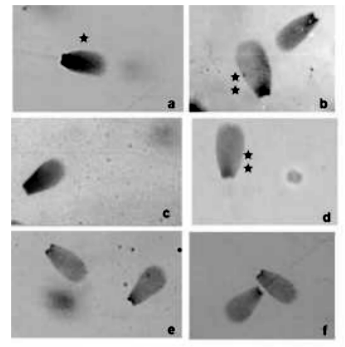
A total of 5250 sperm nuclei from 35 animals (150/animal) were analyzed under light microscopy (x 1000). Light microscopic observations of bovine spermatozoa, stained with the Feulgen reaction, allowed discriminating three different nuclear subpopulations according to the staining intensity, size and shape: normal, large and diploid. Normal nuclei had the morphology more frequent and homogeneous in the population. Large nuclei were wider and longer than normal. Diploid nuclei had a shape similar to that of normal nuclei, were a little larger, and had a slightly broader post-acrosomal zone (Figure 1).
The frequency of normal, large and diploid sperm nuclei showed no significant differences (P>0.05) between fertile bulls and bulls with low fertility and total infertility. Fertile bulls were the 43.3% of the studied animals, bulls with low fertility the 26.7% and bulls with total infertility the 30.0%.
The mean frequency of normal spermatozoa was 92.80% with a range between 84.90% and 97.35%, whereas that of diploid was 0.08%, with a range between 0% and 0.66%, and that of large nuclei was 0.27%, with a range between 0% and 1.22%. No crested, rolled, amorphous or tetraploid nuclei or spermatozoa with double flagellum were detected, but some abnormalities such as small, pear-shaped, narrow and round, were observed with a total mean frequency of 6.85 %. Feulgen staining allowed discriminating clearly between diploid and haploid sperm nuclei because the former were notoriously darker than the latter and the coloration of large nuclei was lighter than that of normal nuclei (Figure 1).
The DNA content, Area, MeAbs, MaxAbs and Volume of individual sperm nuclei were determined in 170 nuclei from five bulls by scanning with the cytophotometric technique (Table 1). Sperm nuclei with C-DNA content were named haploid while those with duplicate DNA content (2C-DNA), were named purportedly diploid since their chromosomes were not cytogenetically identified The DNA content of normal and large nuclei presented no significant differences between them, but differed statistically (P< 0.05) from diploid nuclei. Area, MeAbs, MaxAbs and Volume showed statistical differences (P< 0.05) between normal, large and diploid nuclei.
The mean Area was equal to or smaller than 30 µm2 in normal nuclei and greater than 30 µm2 in large nuclei. The mean Area and Volume of large nuclei were, respectively, 51% and 7% greater than those of normal nuclei. The mean MeAbs and MaxAbs of large nuclei were, respectively, 22% and 13% lower than those of normal nuclei. The mean Area of diploid nuclei overlapped with that of normal nuclei. The mean Area, MeAbs, MaxAbs and Volume of diploid nuclei were respectively, 11%, 78 % 103%, and 94% greater than those of normal nuclei. The mean Area value of diploid nuclei overlapped with that of large nuclei. The mean Area of diploid nuclei was 26% lower than the mean Area of large nuclei. The mean MeAbs, MaxAbs and Volume of diploid nuclei were, respectively, 128%, 135% and 81 % greater than those of large nuclei.
Visual and cytophotometric determinations of 170 sperm nuclei (haploid and diploid) had a percentage of coincidence of 98%.
Figure 2 presents examples of 2D representations of DNA distribution. The 2D graphics show level curves that join points in the (x, y) plane with the same absorbance (points with the same DNA content). The observation of the graphics indicates that MaxAbs always occurred in the basal zone of the spermatozoa and decreased towards the nucleus edge. Moreover, the increase of absorption between the nuclear edge and the MaxAb is notably greater in diploid nuclei than in haploid nuclei.
X and Y axes represent distances (µm2) of the principal area. Curves represent absorbance points with the same absorbance. Absorbance values, are directly proportional to the DNA content.
a) haploid nucleus and b) diploid nucleus. Difference in absorbance values between curves = 0.4. Stars indicate zones of MaxAbs.
4. Discussion And Conclusions
Disorders during premeiosis and/or spermatogenesis can produce diploid and large sperm nuclei (Egozcue et al., 2002; Codina-Pascual et al., 2006; Rosenbusch, 2008). During spermiogenesis, disturbed chromatin condensation due to incorrect replacement of histones and/or formation of disulfide bonds could originate some large nuclei that are associated with low fertility or even total infertility (Ferrari et al., 1998; Revay et al., 2009; Chemes & Rawe, 2012; Guthauser et al., 2016).
In the present work C-DNA, Area, MeAbs, MaxAbs and Volume were cytophotometrically determined to understand the characteristics of the DNA content and chromatin distribution in normal, large and diploid bovine sperm nuclei. Cytophotometric determinations of DNA content allowed us to distinguish two subpopulations: haploid and diploid nuclei. The C-DNA value obtained in normal haploid nuclei (3.23±0.04 pg) confirmed previous data (Gregory, 2017). The DNA content of normal and large nuclei did not differ statistically between them, but, differed statistically (P< 0.05) from that of diploid nuclei. This result is in agreement with data obtained previously (Ferrari et al., 1998).
Numerous studies have analyzed the morphometry of normal bovine sperm heads but few have determined nuclear morphometric parameters. In this work, the value of normal nuclear area (23.77µm2) was similar to that obtained by Foote (2003) (26.20µm2). While in both cases it was used the Feulgen reaction as staining, the small difference that was observed between them may be due to inherited differences and/or the different methods used for measuring the size.
The cytophotometric determination of parameters related to chromatin distribution allows interpreting morphological observations done in bovine sperm nuclei. The nuclear sperm Area was significantly different between normal and large nuclei. Other parameters related to chromatin distribution indicated that large nuclei had a significantly lower MeAbs and MaxAbs values and a significantly greater Area and Volume than normal nuclei, suggesting that they had incomplete and/or defective chromatin condensation. This interpretation is in accordance with that observed by different authors (Ferrari et al. 1998; Revay et al., 2009; Guthauser et al., 2016), but Celik-Ozenci et al. (2004) found that large human sperm heads presented a detectable cytoplasmic retention.
In the present work, the mean Area of diploid nuclei was significantly different from that of both normal and large sperm bovine nuclei. But, values of diploid nuclei overlapped with those of normal and large nuclei and it was difficult to recognize diploid spermatozoa by their size by using light microscopy. Celik-Ozenci et al. (2004) analysing human diploid spermatozoa concluded that sperm size and shape are not reliable attributes in the selection of haploid sperm for ICSI.
MeAbs and MaxAbs of diploid spermatozoa were notoriously higher than those of normal spermatozoa (78% and 103% respectively). These values contrast with the Area, which was only 11% greater than that of normal spermatozoa and suggest that the increase in DNA content would be anisotropic, with a preferential direction of growth along the perpendicular axis to the flattening plane of the sperm nucleus (Figure 2). Similar observations have been made in equine spermatozoa (Spirito et al., 2011) and in llama spermatozoa (Giuliano et al, 2018).
Morphological descriptions of diploid sperm nuclei differ between authors. This can be due to the fact that the spermatozoa were originated by different kinds of failures.
For example, if the meiotic arrest is produced at metaphase (I or II) or at telophase (I or II), the distribution and chromatid composition of the chromosomes could differ.
Revay et al. (2010) found that the chromosomes of bull diploid spermatozoa presented two chromatids in a so close association that they were hardly separable by FISH using whole chromosome painting probes. Based on the observations of Revay et al. (2010) and on the characteristics of the chromatin distribution described in the present study we propose that the diploid bovine sperm nuclei, analyzed in the present work, would have the chromosomes distributed in the principal area, conserving the shape and size similar to that of haploid spermatozoa. Furthermore, chromosomes would be constituted by two closely associated chromatids. This association of chromatids in the chromosomes would generate a large increase in the perpendicular direction to the nuclear flattening plane explaining the almost two-fold increase in the MeanAbs and the MaxAbs and the low increase in the Area.
The frequencies of diploid and large nuclei present in ejaculates are in accordance with data presented by other authors in bovine species (Gledhill 1965; Salisbury & Baker 1966; Barth & Oko; 1989. Ferrari et al., 1998; Vieytes et al., 2008; Pauciullo et al. 2009, González, 2013a). To the best of our knowledge comparisons between fertilization rates and frequency of diploid spermatozoa in bovine ejaculates are not present in the literature. In this paper no statistically significant differences in the frequency of both diploid and large sperm nuclei present in the ejaculates of males with high, mean and low fertility were obtained. But, it should be noted that the only bull that exhibited an important increase in the frequency of diploid sperm nuclei (8.25 times greater than the mean) had low fertility. Vieytes et al. (2008) and Guttenbach et al. (1997) observed similar cases in bovine and human spermatozoa respectively. Although those frequencies were very low, we can presume an "iceberg effect". The meiotic problems that generate diploid spermatozoa could produce other chromosome alterations such as aneuploidies, reducing semen fertility (Egozcue et al., 2000, Godo et al., 2013). It would be interesting that in the future similar cases would be analysed in order to confirm the range of variation of diploid sperm in semen with different fertility levels.
The Feulgen reaction showed to be a simple, non-invasive and low-cost method and our results indicate a 98% of coincidence between cytophotometric determinations and visual light microscope observations. The results obtained in the present work as well as results previously obtained in horses (Spirito et al. 2011), boars (González et al. 2013b) and llamas (Giuliano et al. 2018) allow us to conclude that the Feulgen reaction would be a reliable assay to recognize diploid and large nuclei using light microscopy and should be incorporated in the routine evaluation of species with commercial interest.
Acknowledgements The authors are deeply grateful to the Universidad de Buenos Aires for financial support (UBACyT Projects: 200201001 00811 & 20020130100648BA).
References
- Barth, A. D. & Oko, R. J. (1989). Abnormal Morphology of Bovine Spermatozoa. Iowa State Press / Ames. 1st ed. pp. 3-279.
- Celik-Ozenci, C., Jakab, A., Kovacs, T., Catalanotti, J., Demir, R., P. Bray-Ward, Ward, D. & Huszar, G. (2004). Sperm selection for ICSI: shape properties do not predict the absence or presence of numerical chromosomal aberrations. Human Reproduction, 19, 2052-2059.
- Chemes, H. E. & Rawe, V. Y. (2003). Sperm pathology: a step beyond descriptive morphology. Origin, characterization and fertility potential of abnormal sperm phenotypes in infertile men. Human Reproduction Update, 9, 405-428.
- Chemes, H. E. & Sedo, C. A. (2012). Tales of the tail and sperm head aches changing concepts on the prognostic significance of sperm pathologies affecting the head, neck and tail. Asian Journal of Andrology, 14, 14-23.
- Codina-Pascual, M., Navarro, J., Egozcue, J. & Benet, J. (2006). A human tetraploid pachytene spermatocyte as the possible origin of diploid sperm: a case report. Human Reproduction, 21, 1795-1797.
- Di Rienzo, J., Casanoves, F., Balzarini, M. G., Gonzalez, L,, Tablada, M. & Robledo, C. W. (2015). InfoStat versión 2015. Córdoba, Argentina: Grupo InfoStat, FCA, Universidad Nacional de.Córdoba. Retrieved from: http://www.infostat.com.ar
- Enciso, M.,Cisale, H., Johnston, S.D., Sarasa, J., Fernández, J.L. & Gosálvez, J. (2011). Major morphological sperm abnormalities in the bull are related to sperm DNA damage. Theriogenology, 76, 23-32.
- Egozcue, S., Blanco, J., Vendrell, J. M., García, F., Veiga, A., Aran, B., Barri, P.N., Vidal, F. & Egozcue, J. (2000). Human male infertility: chromosome anomalies, meiotic disorders, abnormal spermatozoa and recurrent abortion. Human Reproduction Update, 6, 93-105.
- Egozcue, S., Blanco, J., Vidal, F., & Egozcue, J. (2002). Diploid sperm and the origin of triploidy. Human Reproduction, 17, 5-7.
- Ferrari, M. R., Spirito, S. E., Giuliano, S. M. & Fernández, H. A. (1998). Chromatin cytophotometric analysis of anormal bovine spermatozoa. Andrologia, 30, 85-89.
- Foote, R. H. (2003). Effect of processing and measuring procedures on estimated sizes of bull sperm heads. Theriogenology, 59, 1765-1773.
- Giuliano, S., Cisale, H. & Ferrari, M.R. (2018). Morphology, morphometry and chromatin distribution in llama sperm nuclei. Reproduction in Domestic Animals 53, 502–511
- Gledhill, B. L. (1965). Cytophotometry of presumed diploid bull spermatozoa Nordisk Veterinaermedicin, 17, 328-335.
- Godo, A., Blanco, J., Vidal, F. & Anton, E. (2013). Accumulation of numerical and structural chromosome imbalances in spermatozoa from reciprocal translocation carriers. Human Reproduction, 28, 840-849.
- González L. Incorporación de las técnicas de evaluación del núcleo espermático en el control de calidad del semen bovino congelado (2013a) Tesis Magister en Biotecnología. Universidad de Buenos Aires (UBA). Biblioteca de la Facultad de Farmacia y Bioquímica (UBA).
- González, L.O., Fischman, M.L., Boquet, M., Acerbo, M..C., Miguez,M.S., Cisale, H.O., Ferrari, M.R. (2013b). Boar semen: complementary techniques for its evaluation. InVet, 15, 37-46.
- Gregory, T.R, 2018. Animal Genome Size Database. http://www.genomesize.com
- Guthauser, B., Pollet-Villard, X., Boitrelle, F. & Vialard, F. (2016). Is intracouple assisted reproductive technology an option for men with large-headed spermatozoa? A literature review and a decision guide proposal. Basic and Clinical Andrology 26, 8.
- Guttenbach, M., Martinez-Exposito, M. J., Michelmann, H. W., Engel, W. & Schmid, M. (1997). Incidence of diploid and disomic sperm nuclei in 45 infertile men. Human Reproduction, 12, 468-473.
- Hardie, D. C., Gregory, T. R. & Hebert, P. D. (2002). From Pixels to Picograms A Beginners' Guide to Genome Quantification by Feulgen Image Analysis Densitometry. Journal of Histochemistry & Cytochemistry, 50, 735-749.
- Levek-Motola, N., Soffer, Y., Shochat, L., Raziel, A., Lewin, L. M. & Golan, R. (2005). Flow cytometry of human semen: a preliminary study of a non-invasive method for the detection of spermatogenetic defects. Human Reproduction, 20, 3469-3475.
- Pauciullo, A., Nicodemo, D., Castello, A., Cosenza, G., Ramunno, L., Rubes, J. & Di Berardino, D. (2009). Chromosomal unbalancements in sperm and oocytes of two Italian cattle breeds as determined by dual color fluorescent in situ hybridization (FISH). Italian Journal of Animal Science, 8, 123–125.
- Pogany, G. C., Corzett, M., Weston, S. & Balhorn, R. (1981). DNA and protein content of mouse sperm: implications regarding sperm chromatin structure. Experimental Cell Research, 136, 127-136.
- Revay, T., Nagy, S., Kopp, C., Flyck,t A., Rens, W., Rath, D., Hidas, A., Kovacs, A., Johannisson, A., Rodriguez-Martinez, H. & Andersson, M. (2009). Macrocephaly in bull spermatozoa is associated with nuclear vacuoles, diploidy and alteration of chromatin condensation. Cytogenetic Genome Research, 126, 202–209.
- Revay, T., Kopp, C., Flyckt, A., Taponen, J., Ljäs, R.., Nagy, S. & Taylor, J. F. (2010). Diploid spermatozoa caused by failure of the second meiotic division in a bull. Theriogenology, 73, 421-428.
- Rosenbusch, B. E. (2008). Mechanisms giving rise to triploid zygotes during assisted reproduction. Fertility and Sterility, 90, 49–55.
- Salisbury G.W. & Baker, F.N. (1966). Nuclear Morphology of Spermatozoa from Inbred and Linecross Hereford Bulls. Journal of Animal Science, 25, 476–479.
- Spirito, S., Campi, S., Boquet, M., Fernández, H. & Ferrari, M. (2011). Equine sperm nuclei with different ploidy levels: relationship between the nuclear DNA content and the nuclear area. Andrologia, 43, 248-253.
- Vieytes, L., Cisale, H. & Ferrari M. (2008). Relationship between the nuclear morphology of the sperm of 10 bulls and their fertility. Veterinary Record,163, 625-9.